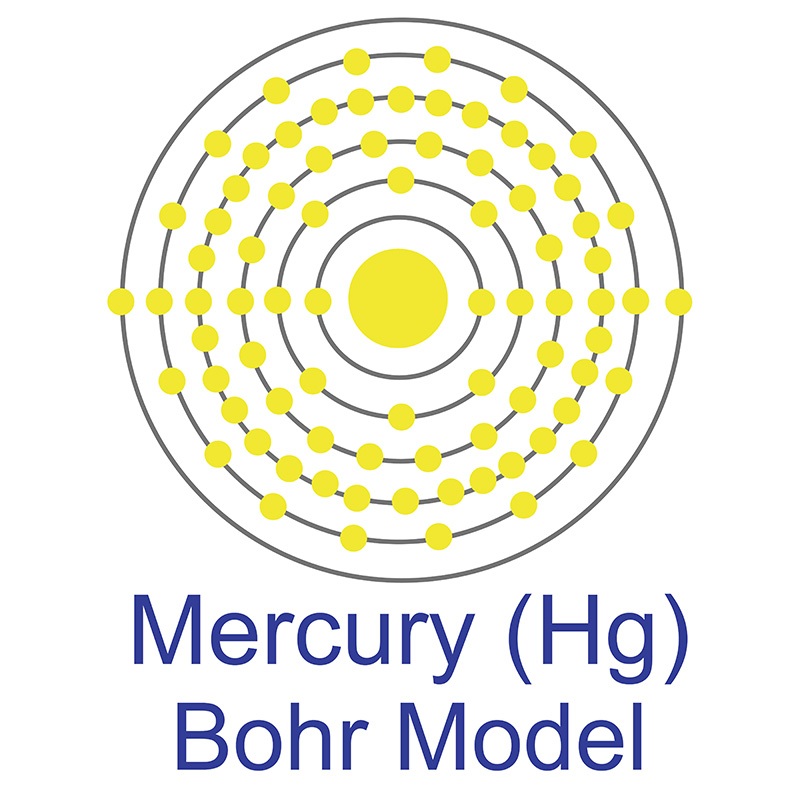
Mercury 206 Electrons
Mercury 200 Lead 206 3 Li 1 5 3 7 3 8O 2 12 g 2 14 Si 4 16 2 20 Ca 2 29 Cu 1 30 n 2 31 Ga 3 35 1 48 Cd 2 53 1 80Hg 2 82 Pb 4. 6 1008664 u 6 1007276 u 6 000054858 u 12000 u.
Mercury Telluride American Elements
We can compare the boiling points of two polar molecules HBr and HCl.

Mercury 206 electrons. Geneous electron transfer from an electrode. Comprehensive data on the chemical element Lead is provided on this page. However its bp189K is lower than that of the less polar HBr 206 K.
Three types of radiation are produced. There are seven stable isotopes of mercury 80 Hg with 202 Hg being the most abundant 2986. Therefore each atom has 80 each of protons and electrons.
An electrode is an electrical con-ductortypicallyplatinumgold mercuryorglassycarbonThrough use of an external power source such as a potentiostat voltage can be applied to the electrode to modulate the energy of the electrons in the electrode. With a standard atomic weight of circa 1008 hydrogen is the lightest element on the periodic table. They also tell you that hydrogen atoms have 12.
Lithium has 3 protons 4 neutrons and 3 electrons. EcellEreduction Eoxidation G 965nEcell G in kJ Ecell E 00592nlog Q log K nE00592 Mol e A time sec96500 time sec mol e 96500current in A t t. Air saturated with mercury vapor at 20C contains a concentration that exceeds the toxicity limits.
The longest-lived radioisotopes are 194 Hg with a half-life of 444 years and 203 Hg with a half-life of 46612 days. The strongest oxidizing agent in the table is MnO 4-aq 8Haq 5e- Mn2aq 4H 2O 151 V Cr 2O 7 2-aq 6e- 2Cr3aq 7H 2O 13V. Use the following list of half-reactions to answer the question.
But what is the driving force for this process. Pure oxygen becoming oxide ion ____ 10. The exact number of neutrons a mercury atom would have depends on its isotopic form but for the.
Including scores of properties element names in many languages most known nuclides of Lead. 199 Hg and 201 Hg are the most often studied NMR-active nuclei having spin quantum numbers of 12. 3 The energy associated with a certain energy level increases with the increase of its distance from the nucleus.
Its monatomic form H is the most abundant chemical substance in the Universe constituting roughly 75 of all baryonic mass. Carbon has 6 protons 6 neutrons and 6 electrons. Now apply the E mc 2 equation to calculate nuclear binding energy.
Mercury is commonly known as quicksilver and was formerly named hydrargyrum. It acts as a cumulative poison and dangerous levels are readily attained in air. Helium has 2 protons 2 neutrons and 2 electrons.
53 Iodine I 206 109 67 54 Xenon Xe 62 55 Caesium Cs 181 56 Barium Ba 149 57 Lanthanum La 1172 58 Cerium Ce 115 101 59 Praseodymium Pr 113 99 60 Neodymium Nd 143 8 1123 61 Promethium Pm 111 62 Samarium Sm 136 7 1098 63 Europium Eu 131 1087 64 Gadolinium Gd 1075 65 Terbium Tb 1063 90 66 Dysprosium Dy 121 1052 67 Holmium Ho 1041 68 Erbium Er 103. All atoms and molecules possess LDF since they all possess electrons. Melting Point-3887 C 23428 K -37966 F Boiling Point35658 C 62973 K 673844 F Number of ProtonsElectrons80.
Table 1 shows decay products and associated energy in MeV million electron volts and the type of particle emitted from the decay. The atomic number of mercury is 80. Skip to page content.
4 An electron jumps from a lower energy level to a higher energy level by absorbing energy. Figure 2 shows the decay from Mercury 206 to Thallium 206 to Lead 206. Once mercury is absorbed it has a very low excretion rate.
Electrons as reactants d. The products are a succession of other radioactive isotopes culminating in the stable isotope lead-206. A major proportion of what is absorbed accumulates in the kidneys neurological tissue and the liver.
HCl is more polar with a larger dipole moment. Protons Neutrons and Electrons of all the Elements. The first decay produces thorium-234.
So mercury has 80 protons and 80 electrons. Beta particles electrons blue gamma radiation orange and alpha particles red and yellow. The chemical symbol for Mercury is Hg.
Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structureThe chemical symbol for Hydrogen is H. Methyl mercury derived from eating fish is readily absorbed in the gastrointestinal tract and because of its lipid solubility can easily cross both the placental and blood-brain barriers. Key Equations Given for Test.
Reactant acting as a reducing agent e. Skip to site menu on this page. Periodic Table of Elements Element Lead - Pb.
Mass defect Total mass of individual sub-atomic particles Mass of the atom. Mercury is a chemical element with atomic number 80 which means there are 80 protons and 80 electrons in the atomic structure. The particles in the nucleus are protons red and neutrons yellow.
Lead-206 has been proposed for use in fast breeder nuclear fission reactor coolant over the use of natural lead mixture which also includes other stable lead isotopes as a mechanism to improve neutron economy and greatly suppress unwanted production of highly radioactive byproducts. According to Figure 1 what is the approximate half-life of Radon 222. These values tell you that a magnesium atom has twice the mass of a carbon atom and 24 times more mass than a hydrogen atom.
Hydrogen has 1 proton 0 neutron and 1 electron. Boron has 5 protons 6 neutrons and 5 electrons. Mercury is a virulent poison and is readily absorbed through the respiratory tract the gastrointestinal tract or through unbroken skin.
The number of neutrons depends on the isotope but for Hg-200 the number of neutrons is 200. 12 C has 6 protons and 6 neutrons and 6 electrons. 2 Electrons revolve round the nucleus in specified circular paths called orbits or shells.
Gain of electrons c. Most of the remaining 40 radioisotopes have half-lives that are less than a day. Beryllium has 4 protons 5 neutrons and 4 electrons.

File 206 Electron Shells 01 Jpg Wikimedia Commons
Solved 5a Give The Subatomic Composition For Each Of The Chegg Com

How To Find The Number Of Protons Electrons Neutrons For Hg Mercury Youtube
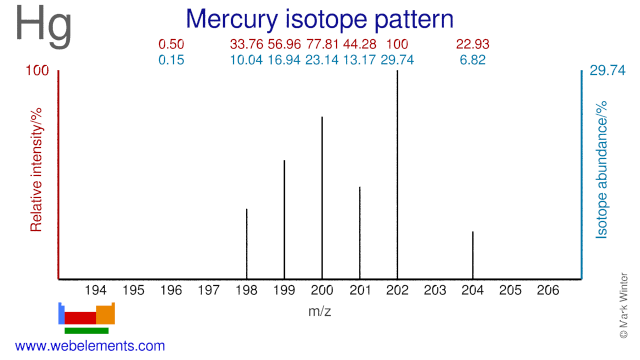
Webelements Periodic Table Mercury Isotope Data

Posting Komentar untuk "Mercury 206 Electrons"