Mercury Symbol And Number Of Neutrons
The five most-abundant isotopes of Mercury have masses from 198 to 202 amu ie. The exact number of neutrons a mercury atom would have depends on its isotopic form but for the most abundant isotope mercury-202 about 30 it would have 202 -.
Mercury Atom Royalty Free Cliparts Vectors And Stock Illustration Image 72753892
Under standard conditions mercury is a shiny heavy silvery liquid.

Mercury symbol and number of neutrons. Figure 233 The symbol Hg represents the element mercury regardless of the amount. Thermometers barometers fluorescent lamps batteries. What is chlorines Atomic number.
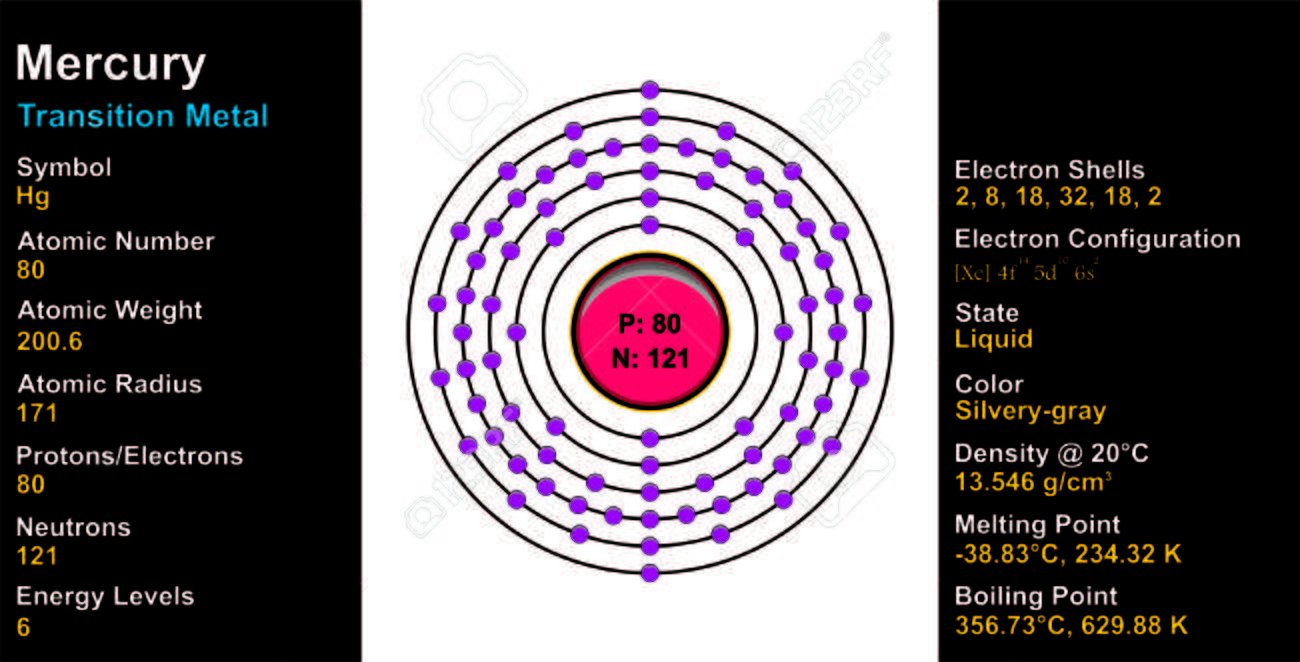
20059 amu Chemical Symbol. Transition Metal Crystal Structure. 80 Number of Neutrons.
To find the number of protons in mercury mercury first locate the element on the periodic table. It could represent one atom of mercury or a large amount of mercury. After the planet Mercury Symbol Origin.
Hg20480 has 80 protons 80 electrons and 124 neutrons. It is an essential constituent of HEMOGLOBINS. 80 Number of Electrons.
We use the same symbol to indicate one atom of mercury microscopic domain or to label a container of many atoms of the element mercury macroscopic domain. A number of neutrons is 124. 20059 amu Melting Point-3887 C 23428 K -37966 F Boiling Point.
For example the symbol for mercury is Hg Figure 3. From the Latin word hydrargyrum liquid silver Uses. Usually gold is created from platinum which has one less proton than gold or from mercury which has one more proton than gold.
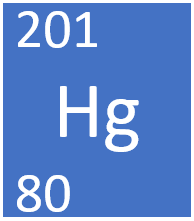
We have a name of this ice Ato is Mercury 204 and our symbol will be to a four for the mass number 80 for the atomic number and its G. Mercury is a chemical element with atomic number 80 which means there are 80 protons in its nucleus. 142 Mercury Date of Discovery.
Mass numbers of typical isotopes of Mercury are 198-202. 20059 amu Number of ProtonsElectrons. 20059 atomic mass units.
Cl Number of neutrons18 Overall charge. The mass of 204 is made up of the protons and neutrons. It number of protons and electrons is 80.
80 Number of Neutrons. We use the same symbol to indicate one atom of mercury microscopic domain or to label a container of many atoms of the element mercury macroscopic domain. Also what has less protons than Mercury.
2320381 Number of ProtonsElectrons. Next find the atomic number which is located above the element s symbol. We use the same symbol to indicate one atom of mercury microscopic domain or to label a.
Protons and Neutrons in Mercury. For example the symbol for mercury is Hg Figure 233. All of these are stable as are two other isotopes with masses of 196 and 204 with 116 and 124 neutrons respectively.
Mercury Number of protons. 198202 neutrons plus the 80 protons of Hg. Mercury atoms have 80 electrons and 80 protons with 122 neutrons in the most abundant isotope.
Known to the ancients Discoverer. Neutral protons and electrons cancel out charges 1. A chemical symbol is an abbreviation that we use to indicate an element or an atom of an element.
For example the symbol for mercury is Hg Figure 213. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Under standard conditions mercury is a shiny heavy silvery liquid.
A chemical symbol is an abbreviation that we use to indicate an element or an atom of an element. A mass number of 200 on an atomic number of 80. 80 Number of Neutrons.
One atom of Mercury has 121 neutrons. Using your three different color beads create a model of the boron atom. 90 Number of Neutrons.
How many neutrons does Mercury 204 have. Mercury was known to ancient Chinese and Hindus before 2000 BC and was found in tubes in Egyptian tombs dated from 1500 BC It was used to forma amalgams of other metals around 500 BC. Rhombohedral Density 293 K.
A metallic element with atomic symbol Fe atomic number 26 and atomic weight 5585. 13456 gcm 3 Color. Thermometers barometers fluorescent lamps batteries Classification.
The mass number is the sum of the mass of the protons plus neutrons. Complete the information for each of the isotopes of mercury. Neutron Number and Mass Number of Mercury.
Known to the ancients Discoverer. The Greeks used mercury in. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N.
The number of neutrons has no effect on the number of electrons. The atomic symbol and the number of protons neutrons and electrons. Herein what is the number of neutrons in mercury.
Well leave us with approximately 120 neutrons. Isotope Protons Neutrons Electrons Isotople Symbol C carbon-14 6 8 6 mercury-200 mercury-201 I 2. 121 Date of Discovery.
35658 C 62973 K 673844 F Number of ProtonsElectrons. Finally we have mercury which has an a top number of 80 mass number two of four for this ice. Look at the Periodic Table of the Elements and locate Boron.
To find the number of neutrons in Mercury you need to first find its atomic number on the Periodic Table of Popular. Since mercury mercury s atomic number is 80 80 Hg Hg has 80 80 protons. What atomic symbol would represent an isotope with 56 protons and 82.
See the example in the first row. Mercury has an atomic number of 80 and a mass number on average mass number 200 059 The atomic number is equal to the number of protons in the nucleus.
Mercury Element Key Stage Wiki

Properties Of Atoms And The Periodic Table Ppt Download

Mercury Protons Neutrons Electrons Electron Configuration
Posting Komentar untuk "Mercury Symbol And Number Of Neutrons"