Mercury 202 Neutrons
D 202 neutrons 198 electrons. Isotope Form Protons Neutrons Electrons 200Hg Atom charge 0 Ion.

Mercury Atomic Structure Stock Image C045 6427 Science Photo Library
80 Number of Neutrons.
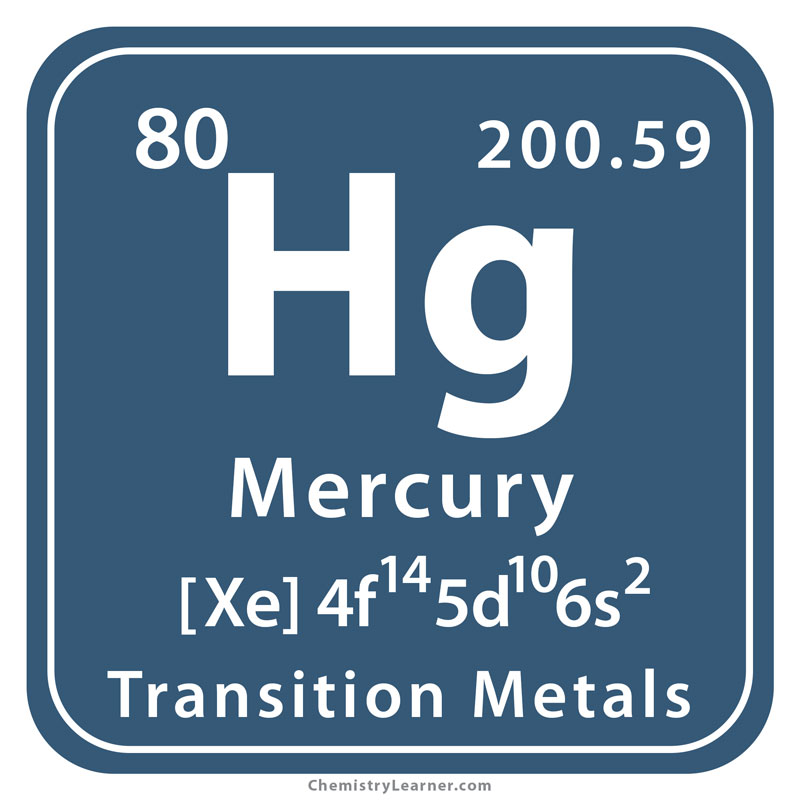
Mercury 202 neutrons. If the mass number of an isotope is known the actual number of neutrons can be calculated. 120 By signing up youll get. A neutral isotope has no charge meaning that the positive and negative charges balance in a neutral isotope.
Neutron number plus atomic number equals atomic mass number. How many electrons does Mercury-202 isotope have. Back to Mercury isotopes list.
Complete the following table showing the number of protons neutrons and electrons for the two most common isotopes of mercury 200Hg and 202 Hg in both the elemental and ion forms. The number of neutrons has no effect on the number of electrons. Most of the remaining 40 radioisotopes have half-lives that are less than a day.
There are seven stable isotopes of mercury 80 Hg with 202 Hg being the most abundant 2986. In the case of mercury the most common isotope is mercury-202. Similarly how many isotopes are in Mercury.
Use the same formula mass number minus atomic number to calculate the number of neutrons. How many neutrons are in Mercury 204. Is Mercury-202 isotope radioactive.
Likewise how many neutrons are in Mercury 202. The mass of 204 is made up of the protons. This element is less reactive than zinc and cadmium it does not displace hydrogen from acids.
Use the equation 202-80122 to find that mercury-202 has 122 neutrons. In the case of mercury the most common isotope is mercury-202. What is the number of electrons in Mercury.
E 198 protons 80 neutrons. Hg20480 has 80 protons 80 electrons and 124 neutrons. 13456 gcm 3 Color.
Besides how many neutrons are in Mercury 200. Mercury 202 Metal is one of over 250 stable Metallic isotopes produced by American Elements for biological and biomedical labeling as. Use the same formula mass number minus atomic number to calculate the number of neutrons.
Hg20480 has 80 protons 80 electrons and 124 neutrons. Use the equation 202- 80 122 to find that mercury -202 has 122 neutrons. Mercury-202 has 122 neutrons in its nucleus.
There are seven stable isotopes of mercury Hg with 202 Hg being the most abundant. Likewise how many protons electrons and neutrons does an atom of mercury 200 have. 171 Hg 172 Hg 173 Hg 174 Hg 175 Hg 176 Hg 177 Hg 178 Hg 179 Hg 180 Hg 181 Hg 182 Hg 183 Hg 184 Hg 185 Hg 186 Hg 187 Hg 188 Hg 189 Hg 190 Hg 191 Hg 192 Hg 193 Hg 194 Hg 195 Hg 196 Hg 197 Hg 198 Hg 199 Hg 200 Hg 201 Hg 202 Hg 203 Hg 204 Hg 205 Hg 206 Hg 207 Hg 208 Hg 209 Hg 210 Hg.
How many neutrons does Mercury-202 isotope have. 120protons neutrons - 50protons. 198 Hg has in its nucleus A 80 protons 122 neutrons.
Mercury is a heavy silvery liquid metallic element belongs to the zinc group. Number of neutrons Mass number - Atomic number 37 - 17 20 neutrons How many neutrons are in the nucleus of the isotopes of tin-120. Mercury-202 is composed of 80 protons 122 neutrons and 80 electrons.
Secondly what is the charge of mercury that has 80 electrons. Mercury 202 Metal Mercury-202 is a stable non-radioactive isotope of Mercury. Use the same formula mass number minus atomic number to calculate the number of neutrons.
In this way how many neutrons are in Mercury 204. 80 electrons green bind to the nucleus successively occupying available electron shells rings. The 2 charge tells us that we have lost two electrons.
C 80 protons 118 neutrons. Here we have 80 protons and 200 - 80 120 neutrons. Hg-196 Hg-198 Hg-199 Hg-200 Hg-201 Hg-202 Hg-204.
The nucleus consists of 80 protons red and 122 neutrons blue. As the atomic mass is determined by the nucleus and the number of protons in an element doesnt change the variation in the number of isotopes alters the weight of an atom. 80 the most common isotope of this element.
What is the mass number of an isotope of mercury that has 122 neutrons. If the mass number of an isotope is known the actual number of neutrons can be calculated. How many protons does Mercury-202 isotope have.
It is used in thermometers barometers and other scientific apparatus. The number of neutrons has no effect on the number of electrons. Mercury-204 is composed of 80 protons 124 neutrons and 80 electrons.
Mercury forms a number of complexes and organomercury compounds. Transition Metal Crystal Structure. Also to know how many.
Isotopes of Mercury click to see decay chain. How many neutrons does the isotope 199 80 Hg have. 20059 amu Melting Point-3887 C 23428 K -37966 F Boiling Point.
Diagram of the nuclear composition and electron configuration of an atom of mercury-202 atomic number. In a neutral isotope the number of electrons equals the number of protons. What is atomic number for Mercury-202 isotope.
In the case of mercury the most common isotope is mercury-202. The longest-lived radioisotopes are 194 Hg with a half-life of 444 years and 203 Hg with a half-life of 46612 days. Use the equation 202-80122 to find that mercury-202 has 122 neutrons.
It is both naturally occurring and a produced by fission. Mercury has seven stable isotopes. So there are 80 2 78 electrons.
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The mass of 204 is made up of the protons and neutrons. 199 Hg and 201 Hg are the most often studied NMR-active nuclei having spin quantum numbers of 12.
Regarding this how many protons does Hg have. Rhombohedral Density 293 K. Is Mercury-202 isotope stable.
Use the equation 202-80122 to find that mercury-202 has 122 neutrons. Mass numbers of typical isotopes of Mercury are 198-202. 35658 C 62973 K 673844 F Number of ProtonsElectrons.
If the mass number of an isotope is known the actual number of neutrons can be calculated. In the case of mercury the most common isotope is mercury -202. B 198 protons 202 neutrons.
Use the same formula mass number minus atomic number to calculate the number of neutrons. In the case of.

Mercury Protons Neutrons Electrons Electron Configuration
Mercury 202 Metal Isotope American Elements
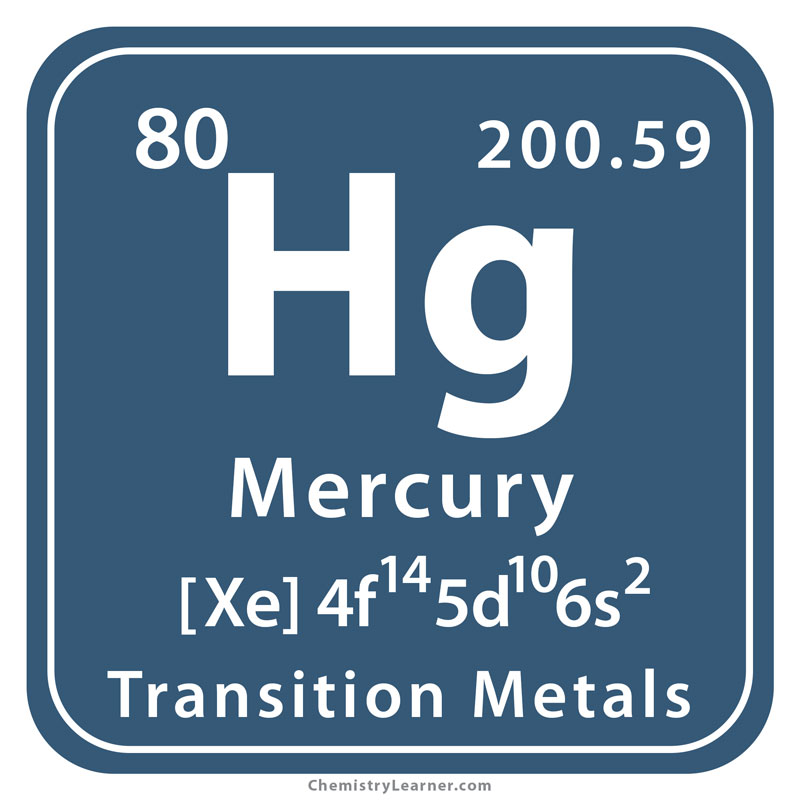
Mercury Definition Facts Symbol Discovery Property Uses

Mercury Protons Neutrons Electrons Electron Configuration
Posting Komentar untuk "Mercury 202 Neutrons"