Mercury(ii) Oxide Decomposes To Its Elements
See the answer. What happens to mercury oxide when heated.

Solved Mercury Ii Oxide A Red Powder Can Be Decomposed By Heating To Produce Liquid Mercury And Oxygen Gas When A Sample Of This Compound Is Decomposed 3 87 Mathrm G Of Oxygen And 48 43 Mathrm G
When heated mercury II oxide decomposes into its elements.

Mercury(ii) oxide decomposes to its elements. When mercuryII oxide a red crystalline solid is heated it decomposes to form liquid mercury and oxygen gas according to the following equation. Why does HgO decompose on heating. Mercury II oxide a red solid decomposes when heated to produce mercury and oxygen gas.
When it is heated it decomposes into mercury. Complete and balance the reaction. MercuryII oxide is an orange highly poisonous substance that is formed when mercury that is exposed to air is heated to 300-350C.
When an electric current is passed through pure water it decomposes into its elements. When the reddish brown mercury II oxide HgO is heated it decomposes to its elements liquid mercury metal metal and oxygen gas. MercuryII oxide a red powder can be decomposed by heating to produce liquid mercury and oxygen gas.
Mercury II oxide a red solid decomposes to form mercury and oxygen gas when heated. MercuryII oxide is a red solid. Equations may be expressed in words.
___HgOs ___Hgl ___O2g unbalanced Balance the equation and determine the mass of mercury that should be formed when 156 g of HgO is heated. MercuryII oxide a red solid decomposes when heated to produce mercury and oxygen gas. MercuryII oxide is a red solid.
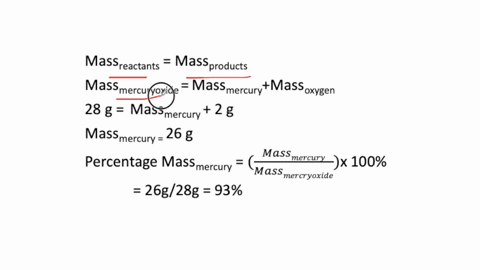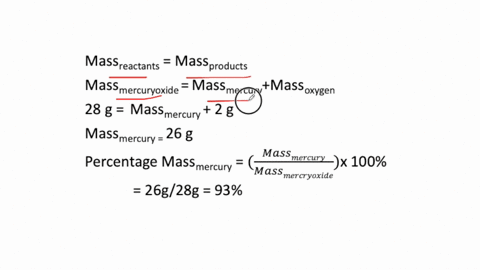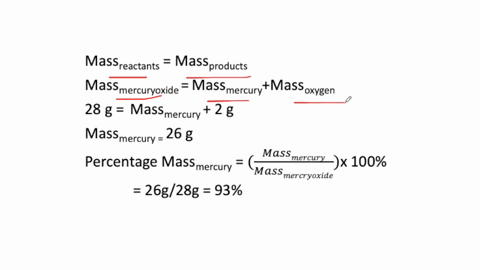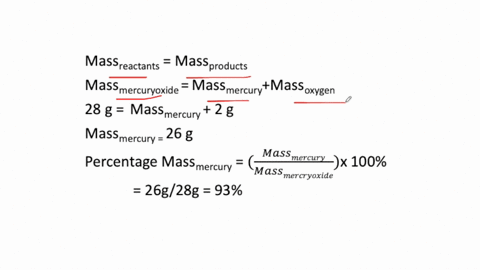
The simplest kind of decomposition reaction is when a binary compound decomposes into its elements. MercuryII oxide decomposes to mercury and oxygen. Above this temperature the free energy of formation of the oxide is positive and the oxide becomes unstable and should decompose into metal and oxygen.
The reactants space those substances v which we start here mercuryII oxide HgO is the reactant. 2Hg O2 is a decomposition reaction in which mercury oxide is decomposed into the element mercury and oxygen gas. What is the silvery material on the walls of the tube.
Mercury II oxide HgO decomposes to form mercury Hg and oxygen O2. At temperatures higher than 400C mercuryII oxide decomposes once again into its consitutent elements. If 429 grams HgO is decomposed to Hg calculate the mass of the pure Hg metal produced.
This reaction should be carried out in a fume hood. Which of the following is a balanced equation for the decomposition reaction that occurs when mercury II oxide decomposes to its elements. The simplest kind of decomposition reaction is when a binary compound decomposes into its elements.
MercuryII oxide a red solid decomposes when heated to produce mercury and oxygen gas. The molar mass of HgO is 21659 gmol. Also a reaction is regarded as a decomposition reaction even if one or more of the products are still a compound.
A metal carbonate breaks down to form a. When reddish-brown mercuryII oxide HgO is heated it decomposes to its elements liquid mercury metal and oxygen gas. If 263 g HgO is decomposed to Hg calculate the mass of the pure Hg metal produced.
Today I perform the decomposition reaction of mercury II oxide also known as mercuric oxide which produces oxygen gas and mercury metal. 2 HgO 2 Hg O2. When the reddish-brown mercuryII oxide HgO is heated it decomposes to its elements liquid mercury metal and oxygen gas.
Mercury II oxide. Using formulas us state this reaction as. The simplest sort of reaction to decomposition is when a binary compound breaks down into its elements.
The simplest kind of decomposition reaction is when a binary compound decomposes into its elements. One now finds that the gas pressure inside the container is 173 atm. The container is then cooled to 25C.
When it is heated it decomposes into mercury metal and oxygen gas. MercuryII oxide is a red solid. 2Hgl O2g-- HgO2s The figure shows the chemical reaction between nitrogen gas to produce ammonia NH3 gas.
2 HgOs 2 Hg l O 2 g we would know that the mercuryII oxide was a solid the mercury was a liquid and the oxygen was a gas when the reaction was carried out. For Teachers for Schools for Working Scholars. 2HgO 2Hg O2.
The molar mass of O2 is 3200 gmol. A sample of mercury II oxide is placed in a 500 L evacuated container and heated until it decomposes entirely to mercury metal and oxygen gas. For example if the equation for the decomposition of mercuryII oxide were written as.
Then I test th. When a sample of this compound is decomposed 387 mathrmg of oxygen and 4843 mathrmg of mercury are produced. MercuryII oxide a red solid decomposes when heated to produce mercury and oxygen gas.
The balanced chemical equation is shown below. Pure mercury II oxide decomposes to its elements. When it is heated it decomposes into mercury metal and oxygen gas.
Write a balanced equation for the decomposition reaction described using the smallest possible integer coefficients. See the answer See the answer done loading.

Solved When The Reddish Brown Mercury Ii Oxide Hgo Is Chegg Com

Solved Ch Ex 20 Mass Of Mercury 3 Attempts Left Check My Chegg Com
Solved When The Reddish Brown Mercury Ii Oxide Hgo Is Chegg Com
Solved Mercury Ii Oxide A Red Powder Can Be Decomposed By Heating To Produce Liquid Mercury And Oxygen Gas When A Sample Of This Compound Is Decomposed 3 87 Mathrm G Of Oxygen And 48 43 Mathrm G
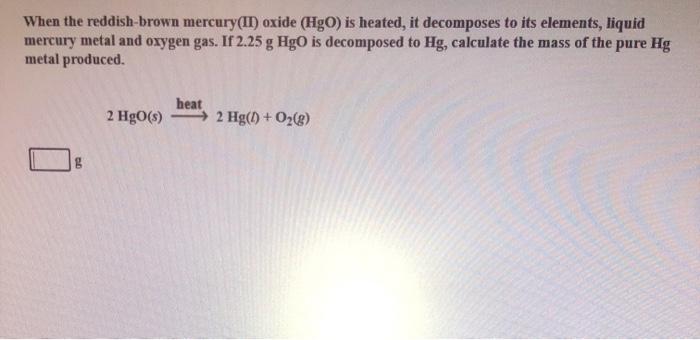
Posting Komentar untuk "Mercury(ii) Oxide Decomposes To Its Elements"