Mercury-200 Neutrons
80 Number of Neutrons. How many neutrons does Mercury have in it.

How To Make Gold From Mercury 7 Steps With Pictures Wikihow
Complete the following table showing the number of protons neutrons and electrons for the two most common isotopes of mercury 200Hg and 202 Hg in both the elemental and ion forms.

Mercury-200 neutrons. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. How many protons does Mercury-200 isotope have. Isotopes are nuclides that have the same atomic number and are therefore the same element but differ in the number of neutrons.
199 Hg and 201 Hg are the most often studied NMR-active nuclei having spin quantum numbers of 12. If the mass number of an isotope is known the actual number of neutrons can be calculated. Other isotopes of mercury are mercury.
The mass number is the sum of the mass of the protons plus neutrons. How many neutrons does Mercury-200 isotope have. How many electrons does Mercury-200 isotope have.
Use the same formula mass number minus atomic number to calculate the number of neutrons. For stable elements there is usually a variety of stable isotopes. Gold isotope of mass 200 Au - PubChem.
Mercury-201 is composed of 80 protons 121 neutrons and 80 electrons. A mass number of 200 on an atomic number of 80. The longest-lived radioisotopes are 194 Hg with a half-life of 444 years and 203 Hg with a half-life of 46612 days.
Atomic mass of Mercury is 20059 u. Use the same formula mass number minus atomic number to calculate the number of neutrons. Mercury Number of protons.
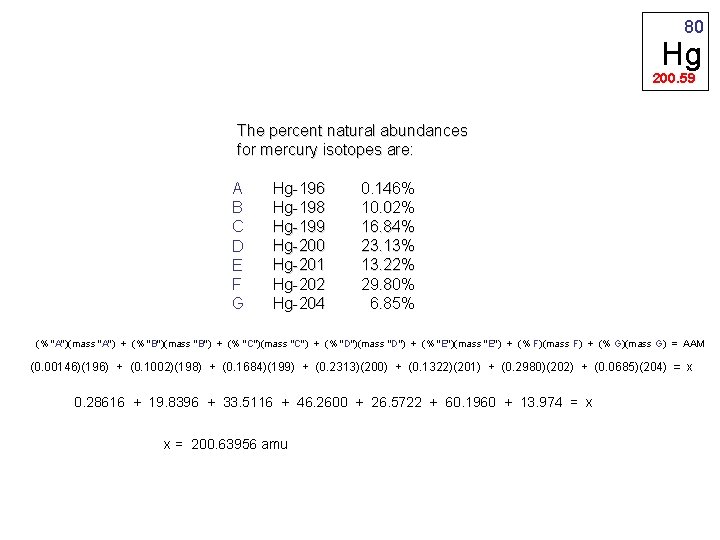
There are seven stable isotopes of mercury 80 Hg with 202 Hg being the most abundant 2986. The element Mercury has 121 neutrons in it. Secondly what is the charge of mercury that has 80 electrons.
Cinnabar aka vermilion mercury sulfide HgS was used as a bright red pigment by the Palaeolithic painters of 30000 years ago to decorate caves in Spain and France. The atomic weight is basically a measurement of the total number of particles in an atom s nucleus including both protons and neutrons. Note that each element may contain more isotopes therefore this resulting atomic mass is calculated from naturally-occuring isotopes and.
Atomic mass of Mercury is 20059 u. Likewise how many protons electrons and neutrons does an atom of mercury 200 have. Simply so how many neutrons are in Mercury 200.
Mercury has an atomic number of 80 and a mass number of 200. Here we have 80 protons and 200 - 80 120 neutrons. Use the equation 202-80122 to find that mercury-202 has 122 neutrons.
In the case of mercury the most common isotope is mercury-202. Likewise people ask how many neutrons does Mercury 202 have. In the case of mercury the most common isotope is mercury-202.
Use the same formula mass number minus atomic number to calculate the number of neutrons. Use the equation 202-80122 to find that mercury-202 has 122 neutrons. Mercury was known to ancient Chinese and Hindus before 2000 BC and was found in tubes in Egyptian tombs dated from 1500 BC It was used to forma amalgams of other metals around.
Atomic Mass of Mercury. 20059 atomic mass units. Question 2 1 point How many protons electrons and neutrons does an atom of mercury-200 have.
Mercury was known to humans as far back as the Ancient Egyptians and its name originates from the alchemy movement after the planet Mercury. Work out the neutrons by taking the protons away from the total mass. Mass numbers of typical isotopes of Mercury are 198-202.
So the first step is to locate the atomic mass on the periodic table and round the value to the nearest whole number. Well leave us with approximately 120 neutrons. If the mass number of an isotope is known the actual number of neutrons can be calculated.
How many neutrons are in one atom of mercury. In the case of mercury the most common isotope is mercury-202. It has the symbol Hg.
Protons 80 electrons 120 neutrons 80 protons 200 electrons 80 neutrons 120 protons 80 electrons 80 neutrons 120 O protons 80 electrons 80 neutrons 200. Most of the remaining 40 radioisotopes have half-lives that are less than a day. Mercury has the atomic number 80 so has 80 protons.
What element has a mass number of 200. It is a Transition metal in Group 12. 120 200 40 80.
Atomic Number of Mercury. The number of neutrons depends on the isotope but for Hg-200 the number of neutrons is 200 - 80 120. Its symbol Hg derives from the Latin Hydragyrum.
Chemistry questions and answers. 80 Number of Electrons. Is Mercury-200 isotope stable.
Neutron number plus atomic number equals atomic mass number. Mercury-202 is composed of 80 protons 122 neutrons. Mercury has an atomic number of 80 and a mass number on average mass number 200 059 The atomic number is equal to the number of protons in the nucleus.
Mass numbers of typical isotopes of Mercury are 198-202. Mercury has an atomic number of 80 and a mass number of 200. What is atomic number for Mercury-200 isotope.
Mercury Hg is a liquid silver coloured metal that has the atomic number 80 in the periodic table. 20059 amu Chemical Symbol. Likewise how many isotopes are in Mercury.
So there are 80 2 78 electrons. Mercury-200 is composed of 80 protons 120 neutrons and 80 electrons. If the mass number of an isotope is known the actual number of neutrons can be calculated.
Use the equation 202-80122 to find that mercury-202 has 122 neutrons. Back to Mercury isotopes list. Isotope Form Protons Neutrons Electrons 200Hg Atom charge 0 Ion.
The 2 charge tells us that we have lost two electrons. Cinnabar would yield up its mercury simply on heating in a crucible and the metal fascinated people because it. Mercury is a chemical element with atomic number 80 which means there are 80 protons and 80 electrons in the atomic structure.
Is Mercury-200 isotope radioactive. How many neutrons are in one atom of mercury. Atomic Mass of Mercury.
The chemical symbol for Mercury is Hg.

Mercury Protons Neutrons Electrons Electron Configuration

How To Make Gold From Mercury 7 Steps With Pictures Wikihow
Intro To Nuclear Chemistry Objective To Use Atomic

Posting Komentar untuk "Mercury-200 Neutrons"