Mercury Relative Atomic Mass
The atomic mass is the mass of an atom. It is a heavy silver-white metal liquid at ordinary temperatures stable in air and water.

Mercury Element In Periodic Table Atomic Number Atomic Mass
The atomic mass or relative isotopic mass refers to the mass of a single particle and therefore is tied to a certain specific isotope of an element.

Mercury relative atomic mass. Hg mercury 20059. Relative Atomic Mass Ar 2 X 185 3 X 187 2 3 186 3 sig fig If the information given is in terms of Abundance the strategy to solve it is the same just take the denominator to be 100 since must add up to 100. Hence the value is said to be relative.
Mercury is a naturally occurring trace metalloid element and known neurotoxin with atomic symbol Hg atomic number 80 and atomic weight 20059. Convert grams HgO to moles or moles HgO to grams. CDC-ATSDR Toxic Substances Portal.
The normal unit of atomic mass has been one-twelfth of the atomic mass of the carbon-12 isotope since 1961. Mercury is an extremely rare element in Earths crust having an average crustal abundance by mass of only 008 parts per million ppm. Atomic mass of Francium Fr 223 223 88.
Atomic Weight amu gmol 1. Ag silver 107868. In 2011 new mass-spectrometric measurements led to A r Hg 2005923.
The atomic mass is the mass of an atom. A_ r frac mass1 times abundance. All other atomic weight values are ratios to the 12C standard value and thus are dimensionless numbers.
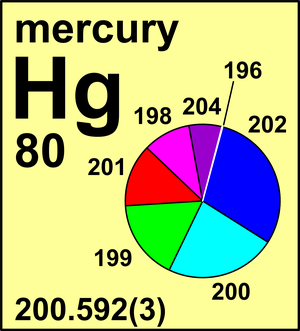
The atomic mass or relative isotopic mass refers to the mass of a single particle and therefore is tied to a certain specific isotope of an element. Published measurements of the isotopic composition of mercury agree remarkably well giving A r Hg values in the range 20058 to 20060. What is the diameter and mass on mercury.
Atomic Mass of Mercury. Mercury Atmosphere Exosphere Surface pressure. One atomic mass unit is equal to 166 x 10.
Atomic mass of Bismuth Bi 20898. Atomic mass of Mercury Hg 20059. 200592 3 Mercury has been known since ancient times.
The mass of Avogadros number of atoms is the atomic mass expressed in grams. In 1989 new measurements of Hg isotope abundances led the Commission to reduce the uncertainty in the standard atomic weight or mercury. Element Mercury Hg Group 12 Atomic Number 80 d-block Mass 200592.
Atomic mass of Radon Rn 222 222 87. The atomic mass of each element is listed below the symbol of the element in the periodic table. The origin of the name comes from the Latin word hydrargyrum meaning liquid silver.
Atomic mass of Mercury is 20059 u. It is a measure which gives the relative mass of an element orcompound - relative to 12C having a mass of 12. Fe iron 55847.
This compound is also known as Mercury II Oxide. 440 K 167 C 590-725 K sunward side Total mass of atmosphere. They are calculated relative to 112 of the mass of a carbon atom which is measured very accurately.
. Molar mass of HgO 2165894 gmol. Atomic mass of Mercury is 20059 u.
Example Exercise 91 Atomic Mass and Avogadros Number. Atomic mass of Polonium Po 209 209 85. Mercury has been used in manufacturing as well as in dental and medical equipment fertilizers and pesticides.
Atomic weight also called relative atomic mass is the ratio of the total weight of the atoms of a chemical substance to a norm. IE in material P is Thus the atomic mass of 12C is 12 u and the atomic weight of 12C is 12 exactly. Atomic mass of Astatine At 210 210 86.
Atomic mass of Thallium Tl 20438. The atomic mass or relative isotopic mass refers to the mass of a single particle and therefore is tied to a certain specific isotope of an element. The atomic weightalso called the relative atomic mass of isotope iE of element E symbol A.
Hydrogen 5000 Potassium 800-1300 Calcium 300-1000. Cu 6355 amu Hg 20059 amu S 3207 amu and He 400 amu. The unit of measure for mass is the atomic mass unit amu.
Sn tin 11871. Atomic mass of Mercury is 20059 u. Since both quantities in the ratio are masses the resulting value is dimensionless.
The atomic mass constant is defined as being 112 of the mass of a carbon-12 atom. Atomic mass of Mercury is 20059 u. Sources facts uses scarcity SRI podcasts alchemical symbols videos and images.
Atomic mass of Radium Ra 226. The relative atomic mass is worked out using the following formula illustrated for two isotopes where the abundances are given in percentage values. Atomic mass of Lead Pb 2072.
Relative atomic mass or atomic weight is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a given sample to the atomic mass constant. Atomic Mass of Mercury. .
The relative atomic masses are introduced to facilitate calculations since the absolute atomic masses of the elements are very small. Note that each element may contain more isotopes therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is the mass of an atom.

The Atom Los Describe The Structure And Properties

Atomic Weight Definition Formula Symbol Atomic Mass Chemistrygod
Atomic Weight Of Mercury Commission On Isotopic Abundances And Atomic Weights

Atomic Number And Atomic Mass Number Definition Examples Questions Mass Number Atomic Number Chemistry Basics
Posting Komentar untuk "Mercury Relative Atomic Mass"