Mercury Ii Nitrate And Potassium Iodide
So you will get a precipitate of white copper I iodide in a brown solution of potassium triiodide. K 2 SO 4 aq BaCl 2 aq-- BaSO 4 s 2 KCl aq Total Ionic Equation.

Properties Of Halogen 06 Reaction Of Potassium Iodide With Silver Nitrate Bos Chemistry Education Resources
Halides are also soluble only the halides of lead mercury and solver are insoluble.

Mercury ii nitrate and potassium iodide. Mercury has the additional property that it is a cumulative poison. 10 mL of 01 M Mercury II chloride solution or you may use mercuric nitrate 20 mL of 2 M Potassium iodide solution. Lead iodide is a yellow.
Solution for mercuryii nitrate and potassium iodide produce mercuryii iodide and potassium nitrate. MercuryII nitrate solution reacts with potassium iodide solution to give a mercuryII iodide precipitate and potassium nitrate solution. Potassium sulfate and barium chloride.
MgNO 3 2 aq CaCl 2 aq - CaNO 3 2 aq MgCl 2 aq Total Ionic Equation. The lead II ion is represented as Pb2 whilst the nitrate ion is NO 3. Pb NO32 aq 2KI aq PbI2 s 2KNO3 aq.
To initial set of a tornado rreaction flask. Repeated low-level exposure can be as bad as a single high-level exposure. Titrations of iodide with mercury II in various acidities using nitrate acetate and chloride as titrants and silver or platinum amalgam as the indicator electrode showed that mercury II nitrate is the best titrant giving 046 V01 ml potential break in comparison with 014 V01 ml of mercury II chloride and 035V01 ml of mercury II.
Strontium nitrate and potassium iodide. Due to the formation of mercuryii iodide it becomes orange. One is mercuryII nitrate and the other is potassium iodide.
Iron water hydrogen iron III oxide 2Fe 3H 2O 3H 2 Fe 2O 3 6. Mercury II chloride or mercury II nitrate. Zinc reacting v hydrochloric mountain produces balloon of hydrogen gas.
Mercury II nitrate solution reacts with potassium iodide solution to give a mercury II iodide precipitate and potassium nitrate solution. They were nitrate oxide and chloride of mercuryII. Hg 2 NO 3 2 2KI Hg 2 I 2 2KNO 3.
529 Copyright 1973 by Academic Press Inc. The reaction can be written as HgNO32 2KI HgI2orange ppt 2KNO3. Two clear solutions are added together.
The chemicals were all of the highest purity avail- able. Mg 2 aq 2 NO 3-aq Ca 2 aq 2 Cl-aq-- Ca 2 aq 2 NO 3-aq Mg 2 aq 2 Cl-aq Net Ionic Equation. Yes it is a double displacement reaction.
It is a very interesting redox reaction. Copper iodide cannot exist. Silver nitrate and ammonium carbonatec.
EXPERIMENTAL METHODS The water used in this investigation was always twice distilled from all glass equipment. Potassium iodide nitric sulfuric and glacial acetic acids. The iodide ions reduce the CuII to CuI and the iodide is oxidised to iodine.
Potassium chlorate potassium chloride oxygen 2KClO 3 2KCl 3 O 2 3. The iodine formed reacts with potassium ions in the reaction mixture to give potassium triiodide. Potassium iodide lead II nitrate lead II iodide potassium nitrate 2KI PbNO 3 2 PbI 2 2KNO 3 4.
The precipitate is yellow in colour and the compound is lead II Iodide. Write the balanced equation and classify the reaction. All nitrates are soluble.
When a colorless systems of leadII nitrate is added to a colorless equipment of potassium iodide a yellow solid dubbed a precipitate is soon produced. When solutions containing mercury II nitrate and potassium iodide were mixed a red-orange precipitate resulted. The chemical equation between mercury II nitrate and potassium iodide as follows.
Mercury I nitrate react with potassium iodide to produce mercury I iodide and potassium nitrate. CopperII sulfate and mercuryI nitrated. To balance the charges we require two nitrate ions per lead II ion and so lead II nitrate is Pb NO32.
When you mix solutions of lead II nitrate and potassium iodide. Lead II Nitrate Potassium Iodide Lead II Iodide Potassium Nitrate. So this is a double displacement reaction.
Write net ionic equations for the reaction if any that occurs when aqueous solutions of the following are mixeda. Mercury ii nitrate reacts with potassium iodide formed mercuryii iodide and potassium nitrate. Solutions for Chapter 4 Problem 52E.
Start with 800 ml of distilled water and add 10 ml of 01 M mercuryII chloride solution. ChromiumIII chloride and sodium hydroxideb. FREE Expert Solution Before balancing the chemical reaction lets first write the chemical formula of each of the compounds involved in the reaction.
Hg NO3 2 2KI ----- Hg I2 2 KNO3 Initial amount of mercury II nitrate 200 g Initial amount of pot View the full answer. The reaction takes place between lead II nitrate and potassium iodide the lead and potassium exchange their anions to give lead iodide and potassium nitrate. A precipitate is a solid product that forms from a reaction and settles out of a liquid mixture.
Iron III oxide carbon iron carbon monoxide Fe 2O 3 3C 2Fe 3CO 5. The mercury builds up in the body and that makes experimenting with mercury extra riskful. Potassium iodide or sodium iodide.
Slowly add a few mls at a time of 2 M potassium iodide. This reaction takes place in a diluted nitric acid.
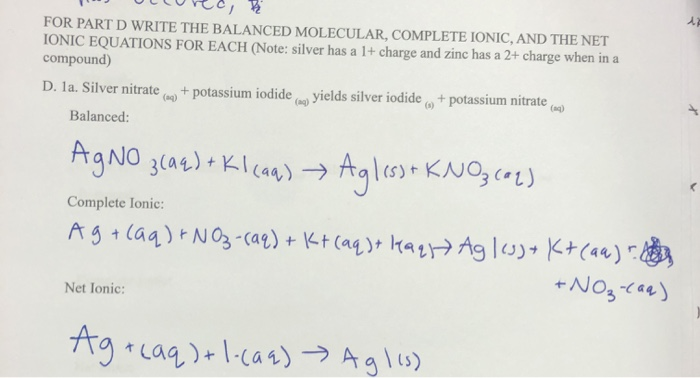
Solved Olcree B For Part D Write The Balanced Molecular Chegg Com
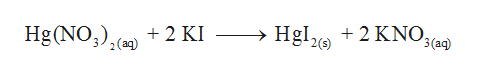
Answered When Solutions Containing Mercury Ii Bartleby

How To Balance Agno3 Ki Agi Kno3 Silver Nitrate Potassium Iodide Youtube
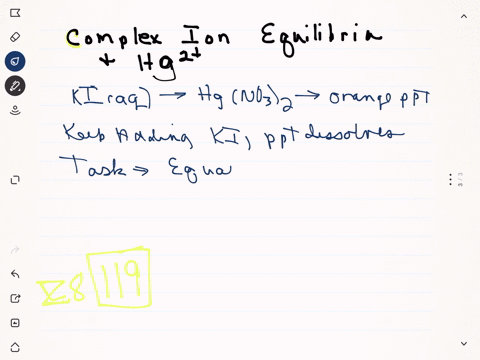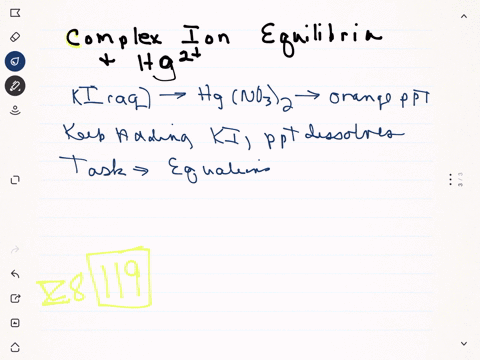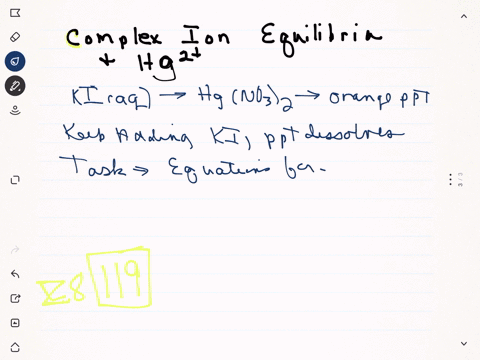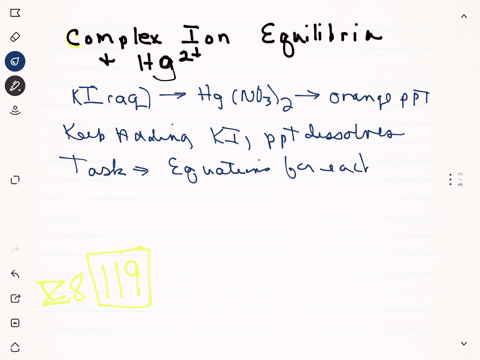
Solved Aqueous Solutions Of Potassium Iodide And Silver Nitrate Are Mixed Forming The Precipitate Silver Iodide
Posting Komentar untuk "Mercury Ii Nitrate And Potassium Iodide"