Mercury Ii Nitrate And Sodium Sulfide
Na2S sodium sulfide 17. AlCl3 aluminum chloride 5.
Solved Please Write The Total Ionic Equation And The Chegg Com
A how many moles of sodium nitrate form from the reaction of 285 mol of sodium.

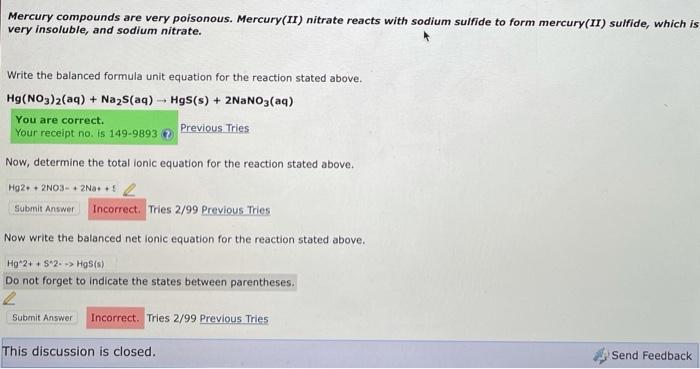
Mercury ii nitrate and sodium sulfide. Symptoms include pain and tightness in chest coughing and difficulty in breathing. H2S Ag1Æ Ag2S H 2. Potassium hydroxide phosphoric acid potassium phosphate water 3KOH H 3PO 4 K 3PO 4 HOH.
Suppose that 200 mL of 001 molL sodium sulphide is used to test a 500 mL sample of water containing 00005 molL mercury nitrate ions. AgCl silver chloride 14. KNO3 potassium nitrate 8.
Solubility is the property of a solid liquid or gaseous chemical substance called solute to dissolve in a solid liquid or gaseous solvent. Calcium chloride and sodium carbonate. Solutions of sodium iodide and lead nitrate are mixed.
What is Soluble and Insoluble. Ammonium sulfide lead II nitrate ammonium nitrate lead II sulfide NH 4 2S PbNO 3 2 2NH 4NO 3 PbS 17. As given amount of mercury II nitrate is more than the required amount.
PbCl2 lead II chloride 7. Solutions of silver nitrate and sodium chromate are mixed. The chemical equation for the reaction of Mercury II nitrate and sodium sulfide follows.
K2SO3 potassium sulfite 13. Ammonium nitrate tin II nitrate iron III phosphate copper I sulfate nickel II acetate mercury II carbonate lead IV hydroxide copper I dichromate copper II chlorate iron II sulfate mercury I perchlorate potassium chlorate tin II sulfate aluminum permanganate lead II nitrate magnesium phosphate copper I dihydrogen phosphate. So it is considered as an excess reagent.
A Write a balanced molecular equation of this reaction. B Determine the theoretical yield of. Hydrogen sulfide is bubbled through a solution of silver nitrate.
Lead IV nitrate sodium sulfate leadIV sulfate sodium nitrate PbNO 3 4 aq 2 Na 2 SO 4 aq PbSO 4 2 s 4 NaNO 3 aq 9. Recently one months production of sodium hydroxide in the US was 221 billion pounds. An excess of sodium hydroxide solution is added to a solution of magnesium nitrate.
An excess of sodium hydroxide solution is added to a solution of magnesium nitrate. CuSO4 copper II sulfate 4. Solutions of sodium iodide and lead nitrate are mixed.
The result is a. MercuryI Nitrate with Sodium Sulfide Play Movie duration 92 seconds size 535 K Here sodium sulfide Na 2 S is added to mercuryI nitrate Hg 2 NO 3 2. Hg 2 aq 2 Cl-aq -- Hg 2 Cl 2 s 3.
Aqueous sodium hydroxide reacts with carbon dioxide gas to yield soluble sodium carbonate and liquid water sodium hydroxide carbon dioxide sodium carbonate water 2 NaOH aq CO 2 g Na 2 CO 3 aq H. The molarity of mercury II nitrate is 0010 M. Write the reaction between solutions of mercury II nitrate and sodium sulfide.
MercuryII Nitrate HgCl2 MercuryII Chloride HgO MercuryII Oxide HgS MercuryII Sulfide HNO2 Nitrous Acid HNO3 Nitric Acid K2C2O4 Potassium Oxalate K2CO3 Potassium Carbonate K2Cr2O7 Potassium Dichromate K2CrO4 Potassium Chromate K2HPO4 Potassium Hydrogen Phosphate K2O Potassium Oxide K2S Potassium Sulfide K2SO4 Potassium Sulfate K3PO4. Ive tried everything but just cant get it. Chronic mercury poisoning can cause kidney mental and nervous disturbances.
I- Pb2 Æ PbI2 4. What is Soluble and Insoluble. The volume of mercury II nitrate is 0050 L.
Reaction of sodium sulfide and mercury ii nitrate. OH- Mg2 Æ MgOH2 3. If ingested toxicity depends on release of t he Hg ion.
Mercury II hydroxide phosphoric acid mercury II phosphate water 3HgOH 2 2H 3PO 4 Hg 3PO 4 2 6H 2O 18. FeO iron II oxide 12. NaOH sodium hydroxide 6.
So 023 moles of sodium sulfide will react with of mercury II nitrate. ZnNO32 zinc nitrate 3. CaCl 2 aq Na 2 CO 3 aq- 2 NaCl aq CaCO 3 s Total Ionic Equation.
The result is a. Solubility is the property of a solid liquid or gaseous chemical substance called solute to dissolve in a solid liquid or gaseous solvent. The density of sodium hydroxide is 2130 gcm³.
HgSO4 Mercury II sulfate is Soluble in water. 1 mole of sodium sulfide reacts with 1 mole of mercury II nitrate. Molar mass of mercuryII nitrate 3247 gmol Moles of mercuryII nitrate mass molar mass 7937 g 3247 gmol 02444 mol Molar mass of sodium sulfide 7805 gmol Moles of sodium sulfide mass molar mass 12026 g 7805 gmol 0.
Ca3PO42 calcium phosphate 15. What mass of precipitate is formed. I would have named it mercuryII nitrate or mercuric nitrate so.
A If a solution containing 74153 g of mercury II nitrate is allowed to react completely with a solution containing 10872 g of sodium sulfide how many grams of solid precipitate will be. Na2S HgNO322NaNO3 HgS As a result of the reaction of sodium sulfide Na2S and mercury ii nitrate HgNO32 produces sodium nitrate NaNO3 mercury sulfide HgS Na2S. The molarity of sodium sulfide is 010 M.
How many cubic kilometers were produced. MercuryI Nitrate with Sodium Sulfide Play Movie duration 92 seconds size 535 K Here sodium sulfide Na 2 S is added to mercuryI nitrate Hg 2 NO 3 2. Mn NO32 Manganese II Nitrate is Soluble in water.
BaOH2 barium hydroxide 16. By Stoichiometry of the reaction. A solution of ammonia is added to a solution of ferric chloride.
The volume of sodium sulfide is 0020 L. Hg 2 NO 3 2 aq 2 HCl aq -- Hg 2 Cl 2 s 2 HNO 3 aq Total Ionic Equation. Acute poisoning can result from inhaling dust concentrations of 12-85 mgm 3 in air.
Hg 2 2 aq 2 NO 3-aq 2 H aq 2 Cl-aq-- Hg 2 Cl 2 s 2 H aq 2 NO 3-aq Net Ionic Equation. Hydrogen sulfide is bubbled through a solution of silver nitrate. Upon mixing 0050 L of 0010 M aqueous solution of mercury II nitrate with 0020 L of 010 M aqueous solution of sodium sulfide a precipitate of mercury II sulfide forms.
Hg 2 F 2.
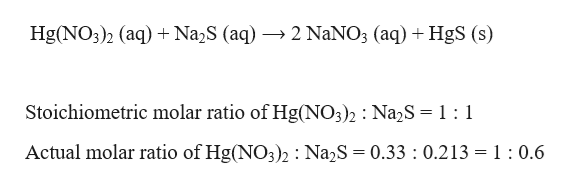
Answered If A Solution Containing 106 19 G Of Bartleby

Solved If A Solution Containing 33 G Of Mercury Ii Nitrate Chegg Com

Solved If A Solution Containing 78 296 G Of Mercury Ii Chegg Com

Molecular Total Ionic And Net Ionic Equations 1
Posting Komentar untuk "Mercury Ii Nitrate And Sodium Sulfide"