Mercury(ii) Sulfide Decomposition
B the surface of aluminum metal undergoes a combination reaction with oxygen in the air. Construct the symbol equation for each reaction in this extraction process.

Chemical Equations Ppt Download
Mercury sulphide chloride chloro-t-butylthiomercury II 2-methylpropene hydrochloric acid and t-butyl chloride.

Mercury(ii) sulfide decomposition. A chemical equation has several parts. Using this information the activation. It is represented by the chemical formula HgS.
Equations may be expressed in words. Thus a pale yellow precipitate of the HgCl22HgS composition forms when H 2 S is passed into a solution of HgCl2. 101039b212914k Mercury sulfide or mercury II sulfide is a chemical.
Version 52 Revision Date 10092012 Print Date 03132014 1. It is virtually insoluble in water. The thermal expansion of cinnabar is fairly isotropic.
The following products of decomposition were identified. Symptoms include pain and tightness in chest coughing and difficulty in breathing. Chronic mercury poisoning can cause kidney mental and nervous disturbances.
At 744 K the thermal decomposition of HgS occurs to elemental mercury and sulfur accompanied by a change in mechanism 2. MercuryIISulfide MercuryII Sulfide Reaction type. Extensive sulfide deposits were associated with volcanism in the earliest stages of the Earths history.
Its existence has been disputed. HgS 10HNO 3 Hg NO 3 2 H 2 SO 4 8NO 2 4H 2 O. Decomposition Please tell about this free chemistry software to your friends.
It may be stable below 0 C or in suitable environments but is unstable at room temperature decomposing into. It may be stable below 0 C or in suitable environments but is unstable at room temperature decomposing into. And iv the CambrianEarly Ordovician.
MercuryII oxide decomposes to mercury and oxygen. Its existence has been disputed. Using formulas we state this reaction as.
B The surface of aluminum metal undergoes a combination reaction. Mercury II sulfide react with nitric acid to produce mercury II nitrate sulfuric acid nitrogen dioxide and water. MercuryI sulfide or mercurous sulfide is a hypothetical chemical compound of mercury and sulfur with elemental formula Hg 2 S.
The decomposition reaction that occurs when solid barium carbonate is heated. Acute poisoning can result from inhaling dust concentrations of 12-85 mgm 3 in air. The rate of thermal decomposition of mercuric sulfide HgS has been measured at temperatures from 265 to 345 C.
The concept of using a thiosulfate dissolutionprecipitation method to stabilize mercury as mercury sulfide has been investigated. The reactants are those substances with which we start here mercuryII oxide HgO is the reactant. MercuryII sulfide red Product Number.
PRODUCT AND COMPANY IDENTIFICATION Product name. Mercury is usually extracted by heating mercury II sulfide in air to form mercury II oxide followed by thermal decomposition of this oxide to give mercury vapour which is then condensed. Not really because you have to achieve very high temperature for cinnabar to fully decompose.
It is possible to identify at least four short epochs of massive sulfide deposition. 1 800-325-5052 Emergency Phone For both supplier and. Section 1 - Chemical Product MSDS NameMercuryII sulfide red 995 Material Safety Data Sheet SynonymVermilion.
If ingested toxicity depends on release of t he Hg ion. Mercury sulfide HgS and mercury selenide HgSe have significantly lower solubilities. A solid and a gas.
Iii the DevonianEarly Carboniferous. These data have been analyzed using a first-order chemical reaction model for the time dependence of the reaction and the Arrhenius equation for the temperature dependence of the rate constant. Mercury sulfide or mercuryII sulfide is a chemical compound composed of the chemical elements mercury and sulfur.
A multi-technique surface study of the mercury ii chalcogenide ion-selective electrode in saline media Analyst 2003 128 742. The sulfide of black mercuryII is normally prepared with hydrogen sulfide by precipitation from an aqueous solution of mercuryII salt. Mercury sulfide or mercurous sulfide is a hypothetical chemical compound of mercury and sulfur with elemental formula Hg 2S.
Solid mercuryII sulfide decomposes into its component elements when heated. The DevonianEarly Carboniferous is the most important period and the CambrianEarly Ordovician less. A solid mercuryII sulfide decomposes into its component elements when heated.
Sigma-Aldrich 3050 Spruce Street SAINT LOUIS MO 63103 USA Telephone. Comparing the solubilities of mercury sulfide and mercury selenide mercury selenide is much less soluble. 1 800-325-5832 Fax.
So is this decomposition technique feasible for producing mercury. Cinnabar from Nevada US. The reaction is first order in the adduct and a mechanism of intramolecular decomposition involving interactions between aliphatic chains and the halogen atoms of mercury.
HgS is dimorphic with two crystal forms. Write balanced chemical equations for the following reactions. 7776 Section 2 - COMPOSITION INFORMATION ON INGREDIENTS.
Nitric acid - concentrated solution. Direct link to this balanced equation. 2 HgO 2 Hg O 2.
Solved Practice Exercise 1 Which Of The Following Reactions Chegg Com

How To Write The Formula For Mercury Ii Sulfide Youtube

Ppt Chemical Reactions Unit Powerpoint Presentation Free Download Id 2043298

Types Of Chemical Reactions Ppt Video Online Download
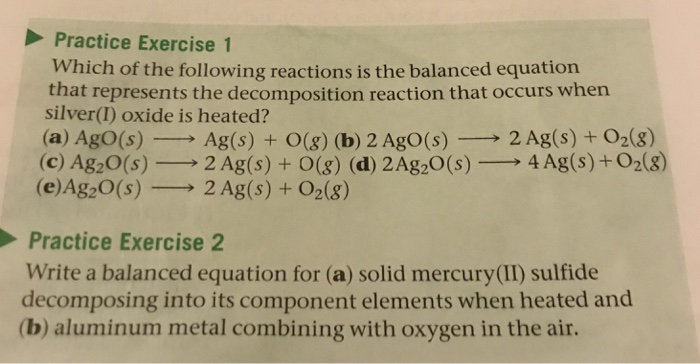
Posting Komentar untuk "Mercury(ii) Sulfide Decomposition"